Printed from acutecaretesting.org
October 2011
POC testing in the emergency department: Strategies to improve clinical and operational outcomes
INTRODUCTION
The majority of hospital emergency departments (ED) in the United States are challenged by a steady increase in patient visits and capacity constraints resulting in overcrowding, delays in providing care and prolonged length of stay (LOS).
Many factors contribute to this problem as outlined in Table I.
TABLE I: Examples of factors contributing to emergency department (ED) overcrowding and prolonged ED length of stay
|
Some factors reflect issues within the ED operation itself while others arise in areas outside of the ED, including delays in radiology, the laboratory and the availability of inpatient beds.
Then there are factors that reflect healthcare policy or societal issues such as the shortage of primary care providers and the presence of large numbers of uninsured and indigent patients without access to traditional medical care.
These patient populations often use the ED for services that would be offered by primary care providers. Because of the diverse origins of these factors, solutions to ED overcrowding require collaboration from a multidisciplinary team to develop an institutional strategy to address operations and patient flow in the ED (Table II).
Ideally the ED operations team is a formal committee that meets regularly to address an ongoing and evolving agenda. As such the team should be a standing hospital committee as opposed to a time-limited task force.
TABLE II: Potential stakeholders and contributors to an emergency department (ED) process improvement team
|
Delays in the laboratory are a common contributing factor to prolonged ED-LOS. Various solutions can be implemented, including reducing delays in specimen transport and reducing the in-laboratory turnaround time.
Alternatively, implementation of point-of-care testing (POCT) for selected tests may improve ED operations by accelerating the evaluation of specific types of patients. This article will describe how POCT can be employed as part of an overall hospital effort to improve clinical and operational outcomes in the ED.
TYPES OF OUTCOMES
Outcomes in a hospital setting can be divided into three types: Medical, financial and operational. Medical outcomes reflect improving the effectiveness of care and can be measured by such metrics as mortality, morbidity or rates of complications.
In practice, medical outcomes can be difficult to measure and often require expensive large-scale studies performed over a significant time frame. Financial outcomes are in principle easy to document. Some intervention either costs more or less than the alternative.
It is generally accepted that POCT usually costs more on a unit-cost basis than central laboratory testing. Determining the unit cost of a laboratory test is fairly straightforward and therefore at first glance it should be easy to calculate the financial outcome for POCT versus using the central laboratory.
In practice, this analysis becomes extremely complex because the cost of the test must be taken in the context of its overall impact on the clinical operation.
For example, consider the case of POCT creatinine testing in radiology. Patients presenting for computed tomography (CT) or magnetic resonance imaging (MRI) scans must have a recent creatinine and calculated estimated glomerular filtration rate available in the medical record before contrast agents can be administered.
However, frequently patients arrive for their scans without the lab test being available [1]. In this case the radiologist has two choices, both of which are suboptimal; either cancel the scan or perform the study without contrast.
If a POCT creatinine is available, the test can be performed immediately and the radiologic studies can proceed without further delay. In terms of unit cost, the POCT creatinine costs several dollars more than a central laboratory test.
However, the impact of the POCT test on cost, revenue and clinical care in the radiology department are significant and far outweigh the incremental cost of the POCT test.
However, it is nearly impossible to perform a reliable cost accounting for the impact of the POCT test because complex interrelated factors must be considered in the analysis such as the payer mix of the patient population, the cost of downtime in the radiology department and a number of other factors.
For this reason financial outcomes with regard to POCT are rarely reliable and inevitably appear contrived. Finally there are operations outcomes. These include such metrics as LOS, ED divert time and patient throughput. Most hospitals already measure these metrics on a continual basis.
For this reason demonstrating an “operations outcome” following implementation of POCT is relatively straightforward.
EMERGENCY DEPARTMENT OPERATIONS
The major tasks of emergency department care include triage, diagnosis, determination of immediate therapy followed by a decision on disposition, either admission, discharge or transfer to another healthcare facility.
The ED operation presents a series of queues starting from the initial patient presentation to the ED and progressing through triage, evaluation, treatment and disposition. In a significant majority of patients, laboratory tests will be requested to assist in the management of the patient [2].
These include common laboratory tests such as chemistry panels and complete blood counts along with more specialized tests such as cardiac troponin, D-dimer and drugs of abuse. Laboratory studies may impact wait times in a number of queues in the ED, thereby having a direct effect on overall ED length of stay.
Several studies have reported on the impact of implementing POCT for a limited menu of routine tests in the ED. Parvin et al [3] implemented a 5-test electrolyte panel and reported no impact on ED-LOS.
Kendall et al [4] studied the impact of a slightly broader menu including blood gases, hematocrit and electrolytes.
While they reported some clinical benefits, again there was no impact on ED operations and efficiency. The most likely explanation for these observations lies in the way common laboratory tests are ordered and are used in the ED in the course of the patient care.
In one study in our ED [2], fully 76 % of patients presenting to the ED received laboratory testing. The average number of tests per patient was 7.7, assuming scoring of standard Medicare panels as one test.
The most common test performed was the complete blood count (66.9 % of patients), followed by the basic metabolic panel (65.4 %), calcium/phosphate/magnesium (51.7 %), hepatic panel (44.1 %) and dipstick urinalysis (35.7 %).
The significant majority of patients received more than one panel. For example, if a patient received a basic metabolic panel, 97.7 % of the time they also had a complete blood count, 77.9 % of the time they had a calcium/phosphate/magnesium panel, 56.9 % of the time a hepatic panel, and so forth.
Therefore simply making one routine test available (electrolytes) has a minimal impact on patient flow because physicians are still required to wait for the results of other routine tests before completing their decisions on management of the patient.
Performing the majority of tests requested on most patients at the POC would require a broad menu on several different instrument platforms.
Furthermore there are no simple, easy-to-use platforms for performing complete blood counts (CBC) in a POC setting.
Unlike basic chemistry and hematology panels, there are some tests that are needed at a key decision point in the course of clinical care.
If the test is positive, one course of action is followed, whereas, if the test is negative, a different course of action is followed. An example is D-dimer testing in the evaluation of venous thromboembolic disease (VTE: includes deep venous thrombosis (DVT) and pulmonary embolism (PE)) (Fig. 1).
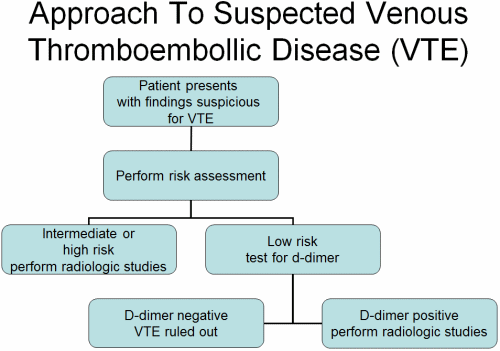
Fig.1
In patients presenting with signs and/or symptoms of VTE the initial step is to perform a risk assessment based on clinical criteria. High- and intermediate-risk patients should all have radiologic studies (usually venous ultrasound (DVT) or computed tomographic scan (PE)).
The majority of patients fall into the low-risk category. In these patients the next step is to perform a D-dimer blood test.
A negative result (most patients) effectively rules out VTE. A positive result raises the index of suspicion and should be followed up by appropriate radiologic studies.
The D-dimer test is therefore central to the evaluation of VTE in low-risk patients. Performance of the test constitutes a queue in the clinical operation that must be completed before the evaluation can proceed. The turnaround time for a D-dimer performed in the clinical laboratory may be as long as 1-2 hours. In contrast, a rapid whole-blood POC D-dimer test takes about 20 minutes.
In our hospital we implemented a POCT whole-blood D-dimer. Following implementation there was a decrease in both the mean and median ED-LOS and a decrease in admissions to the hospital [5].
Various other POC tests have also been shown to improve outcomes in the ED, including rapid cardiac markers [6], B-type natriuretic peptides [7] and urine drugs-of-abuse testing [8]. In each case the key factor was the ability of the test to impact a key clinical decision point or a queue in the operation, enabling the patient to move through the clinical operation more rapidly.
This approach is fundamentally different than simply placing a POCT device in the ED that performs a single test panel that is only a part of the overall general laboratory workup for a particular patient.
Therefore in selecting POC tests for use in the ED the key challenge is to identify those specific tests that have an immediate impact on a clinical decision or reduce wait time in a queue. Only infrequently is POCT required due to an absolute medical necessity. Most tests are implemented mainly to improve efficiency.
MODELS FOR TESTING
Each hospital has its own unique ED and clinical laboratory operations. In some hospitals the clinical laboratory is located immediately adjacent to the ED. In this case there is little need for POCT, albeit the clinical laboratory may utilize devices designed for the POCT to facilitate rapid in-laboratory whole-blood testing.
In other hospitals, the laboratory is located some distance from the ED, perhaps even in a different building on the campus. Many hospitals use pneumatic tube transport systems to facilitate specimen transport from the ED to the laboratory.
Others employ dedicated human couriers. In either case the transport of specimens to the laboratory should, in theory, be rapid. However, in practice this is often not the case. Frequently specimens are collected and then languish in patient treatment rooms or in specimen collection bins at the nursing stations.
In our hospital we studied the total turnaround time for ED specimens being sent to the clinical laboratory, including the preanalytic, analytic and postanalytic turnaround times [6]. Overall the preanalytic turnaround time comprised 42 % of the total, the analytic turnaround time 30 % and the postanalytic turnaround time 28 %.
Although in some cases the ED staff could express samples to the laboratory using our pneumatic tube system, in many cases this did not occur because the nurses were multitasking, including such duties as patient triage, performing clinical evaluations, establishing intravenous lines or administering therapy. Getting the specimen to the clinical laboratory was often not their most immediate priority, resulting in potentially long preanalytic delays.
In 2002 we implemented a point-of-care satellite laboratory located directly in our ED. Over time the menu of tests has expanded to include cardiac markers, D-dimer, urine dipstick and urine pregnancy testing, rapid influenza, RSV and rapid strep A testing.
The facility is staffed by medical technologists and clinical laboratory assistants. This arrangement is expensive but it permits us to perform a wide menu of POC tests without concerns about regulatory compliance, poor quality resulting from operator errors or the performance of tests by untrained personnel.
In order to cost justify this type of model for POCT, the hospital needs to have a large ED sufficient to generate enough specimens to keep the laboratory staff fully deployed.
In smaller hospitals it may make better fiscal sense to train the ED staff to perform the testing themselves. Although this model is much less expensive, its drawbacks include potential issues with regulatory compliance and the fact that it would prove difficult to implement more than a small number of selected POC tests.
CONCLUSION
Implementation of selected point-of-care tests in the ED has been shown to improve ED operations including reducing ED-LOS and altering admission/discharge ratios. The tests should be chosen to impact key decision points or queues within the ED.
Models for implementing POCT include setting up a satellite laboratory staffed by medical technologists or true POCT performed by physicians and/or nurses.
References+ View more
- Lee-Lewandrowski E, Chang C, Gregory K, Lewandrowski K. Evaluation of rapid point-of-care creatinine testing in the radiology service of a large academic medical center: impact on clinical operations and patient disposition. Clin Chem Acta 2011, May 12 (Epub ahead of print).
- Johnson M, Lewandrowski K. Analysis of emergency department test ordering patterns in an urban academic medical center: Can the point-of-care option in a satellite laboratory provide sufficient menu to permit full service testing. Point-of-care 2006; 6: 134-38.
- Parvin C, Lo S, Deuser S, Weaver L, Lewis L, Scott M et al. Impact of point-of-care testing on patient’s length of stay in a large emergency department. Clin Chem 1996; 42: 711-17.
- Kendall J, Reeves B, Clancey M. Point-of-care testing: a randomized, controlled trial of clinical outcome. B Med J 1998; 316: 1052-57.
- Lewandrowski E, Nichols J, Van Cott E, Grisson R, Louissant A, Benzer T, Lewandrowski K. Implementation of a rapid whole blood D-dimer test in the emergency department of an urban academic medical center: impact on ED length of stay and ancillary test utilization. Amer J Clin Pathol, In press.
- Lewandrowski E, Corboy D, Lewandrowski K, Sinclair J, McDermott S, Benzer T. Implementation of a point of care satellite laboratory in the emergency department of an academic medical center: impact on test turnaround time and patient length of stay. Arch Path Lab Med 2003; 127: 456-60.
- Mueller C, Scholer A, Laule-Lilian K, Schindler C, Buser P, Pfisterer M, Perruchoud A. Use of B-type natriuretic peptide in the evaluation and management of acute dyspnea. NEJM 2004; 350: 647-54.
- Lewandrowski K, Flood J, Finn C, Tannous B, Farris A, Benzer T, Lee-Lewandrowski E. Implementation of point-of-care rapid urine drugs of abuse testing in the emergency department of an academic medical center: impact on test utilization and ED length of stay. Amer J Clin Pathol, 2008; 129: 796-801.
References
- Lee-Lewandrowski E, Chang C, Gregory K, Lewandrowski K. Evaluation of rapid point-of-care creatinine testing in the radiology service of a large academic medical center: impact on clinical operations and patient disposition. Clin Chem Acta 2011, May 12 (Epub ahead of print).
- Johnson M, Lewandrowski K. Analysis of emergency department test ordering patterns in an urban academic medical center: Can the point-of-care option in a satellite laboratory provide sufficient menu to permit full service testing. Point-of-care 2006; 6: 134-38.
- Parvin C, Lo S, Deuser S, Weaver L, Lewis L, Scott M et al. Impact of point-of-care testing on patient’s length of stay in a large emergency department. Clin Chem 1996; 42: 711-17.
- Kendall J, Reeves B, Clancey M. Point-of-care testing: a randomized, controlled trial of clinical outcome. B Med J 1998; 316: 1052-57.
- Lewandrowski E, Nichols J, Van Cott E, Grisson R, Louissant A, Benzer T, Lewandrowski K. Implementation of a rapid whole blood D-dimer test in the emergency department of an urban academic medical center: impact on ED length of stay and ancillary test utilization. Amer J Clin Pathol, In press.
- Lewandrowski E, Corboy D, Lewandrowski K, Sinclair J, McDermott S, Benzer T. Implementation of a point of care satellite laboratory in the emergency department of an academic medical center: impact on test turnaround time and patient length of stay. Arch Path Lab Med 2003; 127: 456-60.
- Mueller C, Scholer A, Laule-Lilian K, Schindler C, Buser P, Pfisterer M, Perruchoud A. Use of B-type natriuretic peptide in the evaluation and management of acute dyspnea. NEJM 2004; 350: 647-54.
- Lewandrowski K, Flood J, Finn C, Tannous B, Farris A, Benzer T, Lee-Lewandrowski E. Implementation of point-of-care rapid urine drugs of abuse testing in the emergency department of an academic medical center: impact on test utilization and ED length of stay. Amer J Clin Pathol, 2008; 129: 796-801.
May contain information that is not supported by performance and intended use claims of Radiometer's products. See also Legal info.
Acute care testing handbook
Get the acute care testing handbook
Your practical guide to critical parameters in acute care testing.
Download nowScientific webinars
Check out the list of webinars
Radiometer and acutecaretesting.org present free educational webinars on topics surrounding acute care testing presented by international experts.
Go to webinars








